 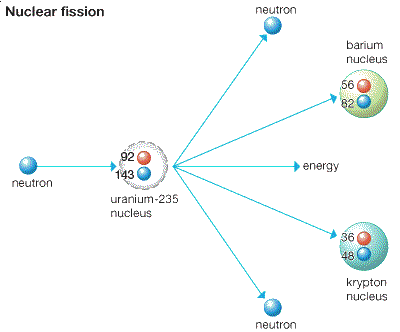
Uranium-238 is an α-particle emitter (occasionally, it undergoes spontaneous fission), decaying through the "Uranium Series" of nuclear decay, which has 18 members, all of which eventually decay into lead-206, by a variety of different decay paths. Much of the internal heat of the earth is thought to be attributable to the decay of uranium and thorium radio-isotopes. The half-life of uranium-238 is about 4.47 billion years and that of uranium-235 is 704 million years, making them useful in dating the age of the Earth. It also suggests that half of the uranium that existed from the formation of the Earth has decayed to other radioactive elements and eventually to stable elements. 
(The US DOE has adopted the value of 0.711 as being their official percentage of 235U in natural uranium.) All three isotopes are radioactive, with small probabilities of undergoing spontaneous fission but preferentially decaying by alpha emission. Naturally occurring uranium consists of three major isotopes: 238U (99.28% abundance), 235U (0.71%), and 234U (0.0054%). There are 27 known isotopes of uranium ranging in atomic weights 217–219, 222–240 and 242, with half-lives of from billions of years to a few nanoseconds. Uranium is weakly radioactive because all naturally occurring ( or primordial) isotopes of uranium ( 238U, 235U and 234U) are unstable, with half-lives varying between 159,200 years and 4.5 billion years. This is high enough to permit a sustained nuclear fission chain reaction to occur, provided other supporting geologic conditions exist. The ore deposit is 1.7 billion years old at that time, uranium-235 constituted about 3% of the total uranium on Earth (0.72% today). In 1972 French physicist Francis Perrin discovered ancient and no longer active prehistoric natural nuclear fission reactors in uranium ore deposits at the Oklo mine in Gabon, West Africa, collectively known as the Oklo Fossil Reactors. The security of those weapons and their fissile material following the breakup of the Soviet Union in 1991 is an ongoing concern. In 1934 research by Enrico Fermi and others eventually led to the use of uranium fission in the first nuclear weapon used in war and later in the peaceful use of uranium as fuel in nuclear power production. An ensuing arms race during the Cold War between the United States and the Soviet Union produced tens of thousands of nuclear weapons that used uranium metal and uranium-derived plutonium-239. Becquerel discovered that uranium exhibited invisible light or rays it was radioactivity. The pure metal was first isolated in 1841 by Eugène-Melchior Péligot, who reduced anhydrous uranium tetrachloride with potassium metal. What Klaproth actually identified was not the pure element but uranium oxide. While experimenting with pitchblende in 1789, he concluded that it contained a new element, which he named after the newly discovered planet Uranus (named after the Greek god of the sky or heaven). The identification of uranium as an element is generally credited to Martin H. In the late Middle Ages, pitchblende was extracted from the silver mines and was used as a coloring agent in the glassmaking industry. Yellow glass with 1% uranium oxide was found in an ancient Roman villa near Naples, Italy. 
when it was used as a yellow coloring agent in ceramic glazes. The use of uranium in its natural oxide form dates back to 79 A.D. Uranium(VI) oxides or "yellow cake" is an intermediate step in the processing of uranium ores. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
